Research Article / Open Access
DOI: 10.31488/HEPH.195
Efficacy and Safety of a Microcurrent Device with High-Frequency Carrier Stimulation for Enhancing Active Attention: A Randomized Sham-Controlled Pilot Trial
Marcus Schmieke, Peter Marmann , Healy GmbH, Kränzlin
*Corresponding author:Dr. Peter Marmann, Healy World GmbH, Office Berlin, Torstrasse 33-35, 10119 Berlin, Germany, Tel: +491718450247.
Abstract
Background: Background: Non-invasive neurostimulation has emerged as a promising tool to enhance cognitive functions, including attention. This study investigates the efficacy and safety of a novel microcurrent stimulation device that utilizes a high-frequency carrier to deliver low-frequency currents aimed at modulating neural activity associated with attention regulation. Methods: A double-blind, randomized, sham-controlled trial was conducted with 131 healthy adult participants aged 20-80 years. Participants were randomly assigned to receive either active stimulation or sham treatment over three weeks twice a day. The active device delivered low-frequency signals (1–100 Hz) modulated onto a high-frequency carrier wave (~5 MHz). Attention performance was measured using a modified digital span test [1,2] before and after the intervention period. Additionally, self-assessments of wellbeing WHO-5 [3,4], assessment of the sense of coherence Soc-9 [5] and assessment of decrease in memory performance related concerns Mymop [6] were performed by the participants as secondary endpoints at the same timepoints. Safety assessments included adverse event monitoring and subjective discomfort reports. Results: Participants of both study groups showed statistically significant improvements in active attention scores (pre/post comparison p < 0.05). The improvement was slightly higher in the active group, but the difference was statistically not significant (p > 0.05). The same could be observed for the secondary endpoints with the exception that the improvement in general wellbeing was significantly higher in the active study group. Only very few Adverse Events related to the study intervention and no serious adverse events were reported, and tolerability was high across both groups. Conclusion: This novel microcurrent device using high-frequency carrier waves to deliver low-frequency stimulation appears to be a safe and effective method for enhancing active attention in healthy adults. Further research is warranted to assess long-term effects and potential therapeutic applications in clinical populations.
Key words: Individualized microcurrent frequencies, high frequency carrier, active attention wellbeing, Device
Introduction
In recent years, there has been growing interest in non-invasive neuromodulation techniques to enhance cognitive functions, including attention, memory, and executive control [7,8]. Attention, particularly active attention-defined as the capacity to sustain focus on task-relevant stimuli while suppressing distractions-is critical to performance in academic, occupational, and everyday settings [9]. Impairments in attention are also central to various neuropsychiatric conditions, such as attention-deficit/hyperactivity disorder (ADHD), depression, and cognitive aging [10].
Microcurrent electrical stimulation (MES) represents a novel class of neuromodulation that uses extremely low-intensity electrical currents, typically in the microampere range, to influence neural activity. Unlike traditional electrical stimulation (e.g., TENS), microcurrent approaches aim to modulate bioelectrical signaling at the cellular level with minimal perceptual discomfort and reduced risk of adverse effects [11]. However, one of the primary limitations of low frequency microcurrents is the challenge of delivering them effectively through the body tissue due to its high impedance. To overcome this, the new developed device now uses a high-frequency carrier wave in the megahertz range to improve signal penetration and delivery efficiency of the therapeutic low-frequency components.
The electric field depends on the gradient of the local electric potential and also on the presence of any time variations of local magnetic potential. For frequencies below around 30 MHz, the effective wavelength of the electromagnetic radiation will be much larger than the dimensions of the various body organs and tissue layers. Calculations of local electric and magnetic potentials can be treated as a near-field problem [12]. Within this study the Healy Highwave device generates approximately 5 MHz as carrier frequency. As consequence the propagation of the original frequencies as generated by the microcurrent device does not take place in the form of current flow or in form of a radiating wave (generated by the carrier device), but as a near-spatial electrical field that interacts with the physical properties of the body [13,14].
Despite the theoretical appeal, empirical evidence supporting the cognitive effects of microcurrent stimulation remains limited. Preliminary studies suggest potential benefits for mood, sleep, pain management and wound-healing [15-17], but its impact on cognitive domains such as attention is underexplored. Moreover, few trials have incorporated rigorous controls, such as sham stimulation, to disentangle placebo effects from true neuromodulatory outcomes.
To address these gaps, we conducted a randomized, double-blind, sham-controlled trial to evaluate the efficacy and safety of a novel high-frequency carrier device for enhancing active attention in healthy adults. We hypothesized that participants receiving active stimulation would demonstrate significant improvements in attention-related outcomes, as measured by a validated neuropsychological test, compared to those receiving sham stimulation. Secondary outcomes included subjective reports of general wellbeing, sense of coherence, specific goal attainment and adverse event monitoring to assess tolerability and safety.
Materials and methods
Design
The study was designed as a two-armed, double blinded sham-controlled study with a three weeks treatment period with a measurement point at the beginning and at the end of the study. A study protocol was finalized before commencement of recruitment. Volunteers were recruited via an existing network of people interested in this type of treatment. After signing the online informed consent form, participants were randomly assigned to one of the two study groups:
Application group (A)
Participants who already use the investigational microcurrent device in daily routine (existing clients) but not already having any experience in using the new high frequency carrier wave generator; application of one Active Attention program designed for elevation of attention two times per day for 3 weeks with current intensity at the microcurrent device between 200 and 1000 µA.
Control group (B)
Same as application group A but deactivated carrier wave (sham application)
Participants
Participants were volunteers who felt moderately mentally exhausted and would profit from some self-help treatment in their general wellbeing, coping with particular issues, such as sleep problems, low affect or lack of energy. They gave informed consent to participate prior to any study specific procedure.
Volunteers were advised to participate only if they would not violate any restrictions for use as given in the Instructions for Use of the Healy device (not intended for pregnant women, persons with epilepsy, heart disease or those with a pacemaker).
Treatment device
For application of frequencies modulated onto a carrier wave the Healy device was connected to the high-frequency applicator via the electrode pins of the Healy device. The coupled devices were attached to the wrist with a bracelet similar to a watch. Starting and controlling the programs is done via the Healy app.
The Healy device uses frequencies from 0.1 Hz to 1 MHz and applies an electrical current between 0 to 4000 μA. The maximum applied voltage is 10 V.
The high frequency applicator generates a carrier frequency of 5 MHz.
Treatment application
Participants assigned to group A applied the frequency program Active Attention. This program is especially designed for improving mental alertness. The program was applied according to a predefined treatment plan two times per day for a total of 3 weeks.
Participants of group B are assigned to use the same program, but the devices of these participants used a negative direct current, which enables the LED of the Healy device to work normally but creates no output signal.
Outcome measures
As primary outcome measure, we used a visual sequence memory test, often used to assess visual working memory, active attention, and short-term memory capacity. This test assesses how well a person can encode, store, and recall a visual sequence after brief exposure. During the presentation phase a sequence of images (shapes, different colored rectangles, photos of faces, are displayed for a short period of time starting with 2 seconds per image and reducing the time stepwise to 0.5 seconds per image). In each sequence 6 images were shown in random order. In the following recall phase, the user was asked to reconstruct the original sequence by clicking the correct order. For each correct position indicated one scoring point was given resulting in a total scoring range from 0 to 72 (12 sequences with 6 pictures).
Due to the deliberately heterogeneous volunteer sample, we opted additionally for one very generic and very individualistic outcome measure. The secondary outcome was the WHO5-Wellbeing scale [18-20], a 5-item scale that has been found to be both very parsimonious, reliable and widely applicable to measure wellbeing as a generic scale. The five items of the scale reflect on the state of the last 2 weeks (cheerful and good spirits, calm and relaxed, active and vigorous, woke up fresh and rested, daily life filled with interesting things) and are rated on a six-point Likert scale (“at no time”, “some of the time”, “less than half of the time”, “more than half of the time”, “most of the time”, “all of the time”). The items can be summed up to yield a sum score ranging from 0 to 25, or, if standardized, on a percentage scale from 0 to 100. Clinically manifest depression is supposed to be present if someone scores less than 50 points, and the population means in European countries are around 70 points. We used the standardized sum score as a main outcome and presented these standardized scores (sum score multiplied by 4).
As an additional secondary outcome we used an individualized score, the Measure Your Own Medical Outcome Profile (MYMOP) Score [21,24]. This is an individually defined measurement system following the generic approach of goal attainment scaling [25]. Individuals are free to define as many – usually up to three – areas of their physical or mental state that they want to see changed. This can be, for instance, sleep, energy and mood in one patient, and mobility, pain and sexual interest in another. This way, everyone can choose their own areas of change. It is initially rated on an 11-point numerical rating scale. The content area is safely stored and implemented in the follow-up measurement for the participant to score once more. We used the (one) most important concern that participants could mention and rate it at the beginning and after 3 weeks of treatment. Besides the assessment of symptom perception at study start and at study completion the participants additionally assessed the improvement of this symptom at the end of the study on a 11-point rating scale lasting from zero = even worsened to 100 = maximal possible improvement.
The third secondary outcome measure was assessed by using the Sense of coherence scale, Leipzig short version. Although SOC is not a direct measure of attention, it's a resilience-related psychological construct. Improvements in active attention and cognitive control often correlate with enhanced coping strategies [26,27]. Increased mental clarity and better perceived control over life circumstances. This instrument consists of 9 items rated on a seven-point Likert scale resulting in a rating range from 7 to 63.
Because our primary outcome was a direct cognitive measure (Visual Sequence Memory), the SOC-9 serves well as a secondary, patient-reported outcome reflecting the psychosocial integration of improved attention.
For the safety assessment, adverse events were queried with an open-ended question in the final survey.
Outcomes were measured by presenting the questionnaires as online questionnaires, as soon as informed consent was received, and then again after three weeks, using an email prompting system that led participants to the online questionnaire.
Ethical statement
This study was conducted in accordance with the principles of the Declaration of Helsinki. It involved healthy adult volunteers and the use of a non-invasive, low risk microcurrent stimulation device intended for general well-being enhancement. The study did not involve any medical intervention, diagnosis, or treatment, and the device used is not classified as a medical device under applicable EU MDR regulations.
In accordance with German research ethics guidelines and institutional policy, no formal ethical approval was required for this type of non-medical, minimal-risk investigation. The study protocol was internally reviewed to ensure compliance with ethical standards, including participant safety, data protection (GDPR), and voluntary informed consent. All participants provided written informed consent prior to enrollment.
Statistical analysis methods
Evaluation of baseline characteristics
To assess the comparability of groups at baseline before the intervention began. Baseline comparability ensures that any observed post-intervention effects are attributable to the intervention rather than pre-existing differences.
Baseline characteristics were evaluated using descriptive statistics including group means and 95% confidence intervals and appropriate statistical tests based on the scale and distribution of the variables.
Continuous variables (pretest scores) were compared using independent samples t-tests (if normally distributed), or Mann–Whitney U tests (if not normally distributed). Normality was assessed using the Shapiro–Wilk test. Homogeneity of variances was assessed with Levene’s test. Alpha level set at 0.05 for descriptive comparisons.
No formal hypothesis testing is required in some regulatory frameworks (e.g., CONSORT), but tests were conducted here for descriptive purposes and to flag potential imbalances.
Pre/Post comparison using paired t-test
A paired samples t-test was used to compare the mean outcome score before and after the intervention within both study groups. This test is appropriate when the same subjects are measured at two time points, the difference scores are approximately normally distributed, and the aim is to detect whether the mean difference is statistically significant.
Hypotheses
Null hypothesis (H₀):
The mean difference between posttest and pretest scores is zero.
H0: μpost−μpre=0
Alternative hypothesis (H₁):
There is a significant change in scores between pretest and posttest.
H1: μpost−μpre≠0
In case the difference scores are not approximately normal distributed, nonparametric test were performed (Z-test and Wilcoxon test).
Group Comparison using Ancova or non-parametric alternatives
In case the required assumptions are fulfilled (Linear relationship between pretest and posttest scores, homogeneity of regression slopes, normally distributed residuals with equal variances) a univariate ANCOVA was performed with posttest scores as the dependent variable, treatment group as the fixed factor, and pretest (baseline) scores as the covariate [28].
Model: Ypost=β0+β1⋅Group+β2⋅Ypre+ϵY
Where:
Ypost: Post-intervention outcome
Group: Intervention (1) vs. Control (0)
Ypre: Baseline pretest value
β1: Estimated treatment effect
In case any of the required assumptions were violated, a non-parametric test was performed.
Software
Statistical analyses were performed using R. A significance level of α = 0.05 (one sided, superiority of the active group was assumed) was used.
Safety assessment
All diseases that were reported in the second survey will be grouped according to the MedDRA system organ classes and listed per application group. Incidences of Adverse Events (AEs) per Organ class and group and incidences of AEs assessed as potentially induced by the study procedure will be compared between the two groups.
Results
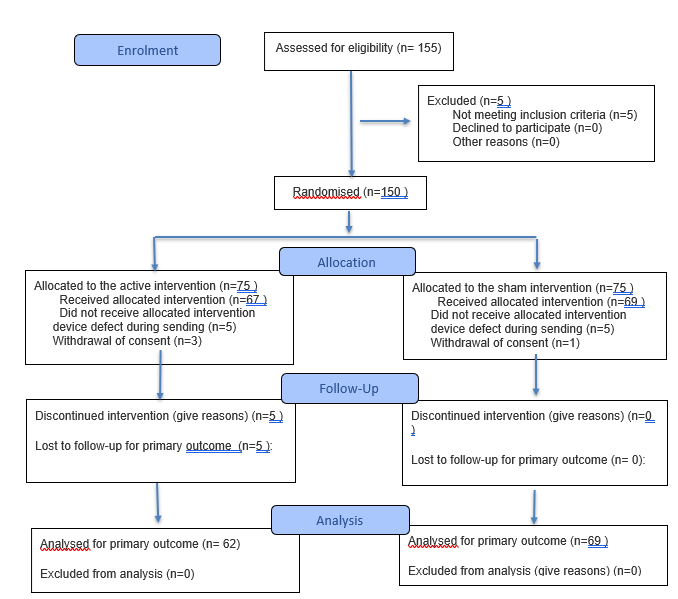
One hundred fifty people consented to the study protocol and were randomly allocated to one of both study groups: active study group (A) or to the sham control group (B), resulting in 75 participants in each study group. Because of device defects during shipment (10 cases) or withdrawal of consent prior to study start (4 cases) in total 14 participants had to be excluded from the study prior to study start. One hundred thirty-six participants completed the first survey. 5 participants terminated the study prematurely; 131 participants completed both surveys and terminated the study according to the protocol.
Since slightly more participants from group A were affected by early terminations, the groups were somewhat unevenly distributed at the end of the study. Group A consisted of 62 participants, while 69 study completers were assigned to group B.
All these 131 completed both questionnaires without missing entries. Consequently, no missing data had to be supplemented with suitable procedures.
Four participants did not complete the first memory test, as consequence these participants were excluded from the evaluation of this parameter.
As can be seen from table 1, the randomization process yielded three quite comparable groups. The majority of the participants, 86%, were female. Due to data-protection concerns age was only collected in rough categories. 70 percent or 80 participants belonged to the middle-aged group between 41 and 60, and nearly half of the rest were older than 60 years or younger than 40 years.
Table 1. Baseline characteristics.
| Active Application (n = 62) | Sham Application (n = 69) | Total (126) | |
|---|---|---|---|
| Gender | |||
| Female | 56 (90%) | 57 (83%) | 113 (86%) |
| Male | 6 (10%) | 12 (17%) | 18 (13%) |
| DNS | 0 (0%) | 0 (0%) | 0 (0%) |
| Age Groups | |||
| 20-40 | 16 (26%) | 8 (12%) | 24 (18%) |
| 41-60 | 38 (62%) | 42 (61%) | 80 (61%) |
| 61-80 | 8 (12%) | 18 (26%) | 26 (20%) |
| >80 | 0 (0%) | 1 (1%) | 1 (1%) |
| Outcome Parameters Baseline | |||
| Visual sequence test Score (0-72) | 45.98 [43.54 -48.43] |
45.23 [42.61 – 47.85] |
45.61 [43.08 – 48.14] |
| WHO-5 Score (0-100) | 41.42 [37.06 – 45.78] |
39.19 [34.82 – 43.55] |
40.30 [35.94 – 44.66] |
| MyMop (0-100) | 77.1 [72.9 -81.3] |
76.4 [72.3 – 80.4] |
76.7 [72.6 – 80.9] |
| SOC-9 | 41.15 [38.84 – 43.45] |
40.53 [38.15 - 42.89] |
40.83 [38.50 – 43.17] |
| Compliance | |||
| Compliant | 43 (69%) | 52 (75%) | 95 (73%) |
| Non-Compliant | 19 (31%) | 17 [25%] | 36 (27%) |
The baseline values of all endpoint parameters differed only marginally in both groups. The respective 95% confidence intervals overlapped and the t-tests for independent samples did not reveal any significant group differences (t = 0.82, p = 0.41 for the visual sequence test, t = 0.52, p = 0.60 for WHO-5; t = 0.001, p = 0.99 for Mymop, t = 0.53, p = 0.60 for SOC-9 for dF 125 each).
Participant’s compliance was determined by the frequency of study programs used during the study period. This is tracked in a cloud and can be read from there. In both groups, compliance, defined as at least 50% of the intended applications performed, was close to 90%. The two groups differed only very slightly (CHIQU-test, p = 0.89).
Gender, Age-Groups, Group (Active application, Sham); absolute frequencies and percentages (per category and group); mean scores for WHO-5, MYMOP scale, Memory Test Score and Sense of coherence SOC-9 [95% Confidence Intervals]; compliance per treatment group as defined as follows: for both treatment groups at least 50% of the stipulated applications are defined as compliant, less than 5 application were defined as non-compliant.
Primary endpoint visual sequence test
The primary efficacy analysis assessed cognitive performance using a standardized memory test at the final study visit. Adjusted mean scores were derived using analysis of covariance (ANCOVA), controlling for baseline memory scores.
Both treatment groups demonstrated statistically significant improvements in visual memory performance, as measured by the Visual Sequence Test from baseline to the final visit (table 2 and figure 1).
Table 2. Results of statistical tests of Pre Post Comparisons of endpoint parameters
| Active application (n = 62) |
Sham application (n = 69) |
||||
|---|---|---|---|---|---|
| Test | Statistic | p | Statistic | p | |
| Visual sequence test | Student | 2.832 | 0.0061 | 2.434 | 0.0181 |
| Wilcoxon | 1.260.500 | 0.002 | 1.429.000 | 0.004 | |
| Z | 34.954 | < .001 | 33.727 | < .001 | |
| WHO-5 Score | Student | 8.782 | < .001 | 7.394 | < .001 |
| Wilcoxon | 1.636.000 | < .001 | 1.829.500 | < .001 | |
| Z | 39.307 | < .001 | 33.727 | < .001 | |
| MyMop | Student | 8.716 | < .001 | 6.873 | < .0011 |
| Wilcoxon | 1.422.000 | < .001 | 1.560.000 | < .001 | |
| Z | 22.022 | < .001 | 20.433 | < .001 | |
| SOC-9 | Student | 6.635 | < .001 | 6.046 | < .001 |
| Wilcoxon | 1.463.000 | < .001 | 1.785.500 | < .001 | |
| Z | 51.215 | < .001 | 47.144 | < .001 | |
Note: For the Student t-test and Z-test, the alternative hypothesis specifies that the mean is different from 0. For the Wilcoxon test, the alternative hypothesis specifies that the median is different from 0.
1: Shapiro-Wilk test indicates deviation from normal distribution
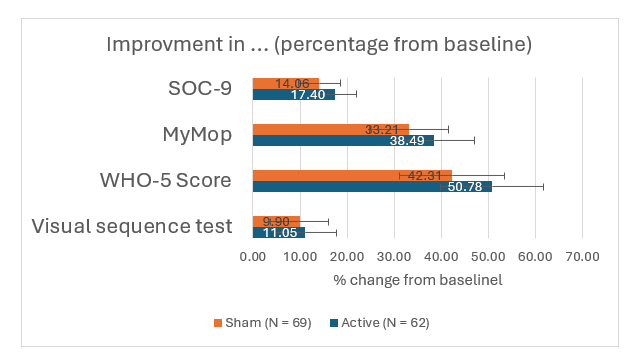
Figure 1:Percentage increase in visual sequence test scores, WHO-5 score, and SOC-9 score, respective percentage decrease in Mymop severity score; Mean values (±95% CI).
In the control group, the mean improvement was 9.90%, with a p-value < 0.02 based on a paired t-test, indicating a significant within-group change.
In the active treatment group, the mean improvement was 11.05%, also statistically significant (p = 0.006, paired t-test).
These findings indicate that both interventions were associated with meaningful gains in visual sequence memory over the course of the study.
The adjusted mean score for the active device group was 51.07, compared to 49.71 in the sham device group (figure 2). The between-group difference in adjusted means was 1.36 points in favor of the active device.
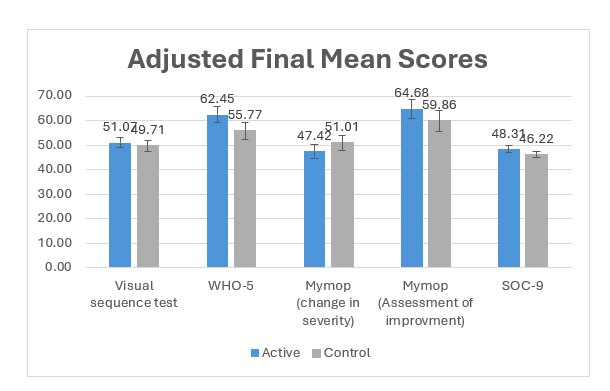
Figure 2:Adjusted means (±95% CI) of the final endpoint by group, estimated using baseline correction.
A one-sided t-test comparing the adjusted means yielded a p-value of 0.27 (table 3), which did not reach statistical significance at the pre-specified alpha level (0.05). These results suggest no statistically significant superiority of the active device over the sham control in improving memory performance at the final time point.
Table 3. Result of statistical tests of group comparisons; one sided t-test for residuals after baseline correction
| DF | t | p | |
|---|---|---|---|
| Visual sequence test | 124 | 0.607 | 0.272 |
| WHO-5 | 129 | 1.899 | 0.030 |
| Mymop Severity | 129 | 0.926 | 0.178 |
| Mymop Application Success | 129 | 1.438 | 0.076 |
| SOC-9 | 129 | 1.472 | 0.072 |
Secondary Endpoints
WHO-5 Well-Being Index
Participants in both the active and sham control groups demonstrated significant improvements in well-being as measured by the WHO-5 Well-Being Index. In the active group, mean WHO-5 scores increased from 10.35 (SD = 4.3) at baseline to 15.6 (SD = 3.9) post-intervention, representing a mean improvement of 5.25 points. In the sham control group, scores increased from 9.8 (SD = 4.5) to 13.94 (SD = 5.4), a mean gain of 4.14 points. After adjusting for baseline differences using ANCOVA, the adjusted mean score was 62.45 in the active group and 55.77 in the control group. The between-group difference was clinically and statistically significant (p = 0.03), suggesting that the active intervention had a superior effect on subjective well-being.
A between-group comparison of post-intervention scores yielded a Cohen’s d of 0.35, indicating a small to moderate effect size in favor of the active treatment. The pooled post-test standard deviation was 4.75, and the between-group difference in post-intervention means was 1.66 points.
These results indicate that both interventions produced clinically meaningful improvements in subjective well-being, with the active intervention yielding additional benefits beyond those attributable to placebo effects or non-specific treatment factors.
MYMOP (Measure Yourself Medical Outcome Profile)
The MYMOP severity score, which reflect participant’s-reported symptom burden, decreased in both groups, indicating improvement:
Active group: 38.49% decrease; Control group: 33.31% decrease
ANCOVA-adjusted means were 47.42 (active) vs. 51.01 (control). The between-group difference was not statistically significant (p = 0.178), indicating that both groups improved similarly in symptom perception. At the final study visit, participants rated the improvement of their primary concern using an 11-point Likert scale ranging from -5 ("even worsened") to +5 ("maximal possible improvement"). The mean improvement score in the active treatment group (N = 62) was 1.47 (SD = 1.88), compared to 5.99 (SD = 2.05) in the control group.
An independent samples t-test revealed a trend toward greater perceived improvement in the active group; however, the difference did not reach statistical significance (t = 1.44 df = 129, p = 0.076).
SOC-9 (Sense of Coherence Scale)
Improvements were also observed in psychological resilience, as measured by the SOC-9:
Active group: 17.40% increase; Control group: 14.06% increase.
The ANCOVA-adjusted mean scores were 48.31 for the active group and 46.22 for the control group. While the trend favored the active treatment, the difference was not statistically significant (p = 0.072), though it may indicate a clinically relevant signal that warrants further investigation.
Safety Evaluation
Overall, new diseases occurred in 31 of the 131 study participants during the course of the study (table 4). The proportion of participants with diseases over the course of the study was slightly higher in the active group than in the control group, but the difference was not significant (Chiqu test p = 0.17). Only two participants per group assumed that there was a connection between the disease and the use of the test device. In the active group, the corresponding diseases were nausea and headaches, in the control group headaches and a contact allergy (pustules at the point where the electrodes touched the skin). The most common adverse event was flu-like infections (15 in total), followed by headaches (3 cases) and joint pain (2 cases).
Table 4. Safety assessment, Occurrence of Adverse Events in the course of the study
| Active (N = 62) | Control (N = 69) | |
|---|---|---|
| Number of AEs | 18 | 13 |
| Participants with AE | 29% | 19% |
| AEs related to application | 2 (Nausea, Headache) | 2 (Headache, Contact allergy) |
| Description of AEs | ||
| Infection | 10 | 5 |
| Dizziness | 1 | |
| Nausea | 1 | |
| Headache | 2 | 1 |
| Joint pain | 2 | |
| Acute stress | 1 | |
| Wound | 1 | |
| Migraine | 1 | |
| Contusion | 1 | |
| Tiredness | 1 | |
| Contact allergy | 1 | |
| Allergy | 1 | |
| Stomach intestinal complaints | 1 | |
| Colon Inflammation | 1 | |
| AEs leading to premature termination | 1 (Headache) | 1(Colon Inflammation) |
| Serious Adverse Envent | 0 | 0 |
Two adverse events (one in each study group) resulted in early discontinuation. In the active group there was a headache, in the control group an intestinal inflammation.
None of the adverse events were serious.
Discussion
This randomized, sham-controlled pilot trial evaluated the efficacy and safety of a high-frequency carrier microcurrent (HFCM) device for enhancing active attention, with memory performance as the primary outcome. While both the active and sham groups demonstrated statistically significant improvements in memory test performance from baseline to post-intervention, no significant or clinically relevant differences were observed between the two groups.
The within-group improvements suggest a potential placebo effect, practice effect from repeated cognitive testing, or non-specific benefits of study participation, such as increased motivation or engagement. These factors are common in cognitive intervention studies and highlight the importance of sham-controlled designs when evaluating novel neuromodulation technologies.
The lack of a between-group difference indicates that, under the conditions of this trial, HFCM stimulation did not produce a measurable additive effect beyond that of sham treatment. Several factors may account for this. The relatively short duration of the intervention, the use of a cognitively healthy sample, and the sensitivity of the cognitive assessments may have limited the detection of more subtle effects attributable to the active stimulation. Additionally, while the frequency program is designed to influence active attention, the specific parameters used in this study may not have been optimal for modulating cognitive networks related to memory.
Future studies should consider using longer stimulation protocols, larger sample sizes, and possibly focusing on populations with attention or memory impairments, where the potential for improvement is greater. Incorporating objective biomarkers such as EEG or neuroimaging could also help clarify whether the HFCM device induces any neural changes that are not captured by behavioral outcomes alone.
In summary, while both groups showed pre/post improvements in memory performance, the absence of a significant difference between active and sham stimulation suggests limited efficacy of the HFCM device for cognitive enhancement in healthy individuals under the tested conditions. These findings underscore the importance of rigorous control conditions and call for further investigation in targeted populations and with optimized stimulation parameters.
In contrast, several secondary outcomes provided preliminary evidence of potential benefit specific to the HFCM intervention. The present findings demonstrate that both the active and sham interventions were associated with significant increases in subjective well-being, as measured by the WHO-5, over the course of the treatment period. The mean improvements in both groups exceeded the minimum clinically important difference (MCID) of approximately 2.5 points (10% of the WHO-5 scale), a threshold commonly used to signify clinically relevant change[29,30].
Crucially, the additional 1.66-point improvement in the active group, corresponding to a Cohen’s d of 0.35, reflects a modest but clinically relevant treatment-specific effect. This finding aligns with prior research indicating that placebo effects can account for a substantial portion of observed improvements in psychological outcomes, especially in interventions focused on mood, stress, and subjective well-being [31,32]. Nonetheless, the added benefit of the active treatment underscores its value beyond expectancy and engagement effects alone.
The WHO-5 is a widely used and well-validated measure of psychological well-being, particularly sensitive to changes in depressive symptomatology and general affective state [33,34]. A post-treatment score above 13 is considered a marker of improved mental health status 34, and participants in both groups reached this benchmark on average by the end of the intervention. However, only the active treatment group exceeded this threshold by a larger margin (15.6 vs. 13.94), suggesting a stronger shift into the range of normative well-being.
While the placebo response observed in the sham group was substantial-a common phenomenon in psychological interventions-it does not diminish the practical importance of the treatment-specific effect. In real-world clinical or public health settings, incremental improvements in well-being of even 1-2 points on the WHO-5 can translate into meaningful reductions in functional impairment, distress, or risk of depression [35].
Additional secondary measures further support this trend
The Measure Yourself Medical Outcome Profile (MYMOP) showed significant improvements in therapy success ratings in both groups. While the between-group difference did not reach conventional statistical significance, it approached significance (p ≈ 0.07), favoring the active group, indicating a potential trend toward perceived therapeutic benefit with HFCM.
The MYMOP symptom severity ratings improved significantly within both groups, but no significant differences between the groups were detected, suggesting that while participants perceived symptom improvement, this may not have been specifically attributable to the active stimulation.
While the active group reported a higher mean improvement (6.47) compared to the control group (5.99), the difference did not reach statistical significance (p = 0.07). However, the observed trend may suggest a potentially clinically meaningful benefit of the active intervention.
The Sense of Coherence Scale (SOC-9) also improved significantly within both groups, with a near-significant between-group difference (p = 0.07) favoring the active intervention. This finding hints at a possible role for HFCM in strengthening psychological resilience or stress-coping capacity.
Taken together, the consistent within-group improvements across all secondary outcomes and the trend-level or significant superiority of the active group in well-being-related measures suggest that HFCM stimulation may have a more robust effect on subjective and emotional dimensions of health than on cognitive performance per se, at least in the short term and in a healthy population.
Importantly, the intervention was well tolerated with no serious adverse events reported, reinforcing the safety of HFCM application under study conditions.
Limitations
These results should be interpreted in light of several limitations. First, the use of a sham control, while valuable for estimating placebo effects, may not fully isolate all non-specific factors (e.g., attention, engagement). Second, the reliance on pre-post effect sizes assumes comparable baseline characteristics and no regression artifacts, although both groups were well-matched at baseline. Third, self-reported outcomes may be subject to response biases; future studies may benefit from including behavioral or biological indicators of well-being.
Conclusion
While the primary endpoint did not reveal a specific cognitive benefit of HFCM stimulation, the consistent pre/post improvements and the emerging between-group differences in well-being and perceived therapy success suggests promising potential in psychological domains. These findings justify further investigation in larger trials with longer follow-up, ideally including populations with reduced baseline well-being or specific cognitive or emotional vulnerabilities. Future research should also explore neurobiological correlations to better understand the mechanisms behind the observed effects.
Abbreviations
AE: Adverse Event; CI: Confidence interval; Hz: Hertz; IMF: Individualized Microcurrent Frequency; IP: Investigational Product; MDD: Major depressive disorder; MedDRA: Medical Dictionary for Regulatory Activities; MES: Microcurrent electrical stimulation; MHz: Mega Hertz; μA= Milli Ampere; MYMOP: Measure Yourself Medical Outcome Profile; PSS: Perceived Stress Scale; SOC: Sense of coherence;; V: Volt; WHO: World Health Organization.
Authorship statement
Dr. Peter Marmann (PM) organized the study, recruited the participants and collected the data. He also analyzed the data and wrote the first draft of the manuscript.
Marcus Schmieke (MS) developed the design of the study and finalized the manuscript.
Sponsoring
The study was sponsored by Healy GmbH, Kränzlin, Germany.
Role of the sponsor
The sponsor helped with recruitment by activating his network of users. The sponsor suggested some aspects of the design, like the two active modes of action, superiority of the active group against the untreated control group.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Conflict of interest
The author has the following conflict of interests: The Author Dr. Peter Marmann is an employee of Healy GmbH, the sponsor of the study.
References
1. Jahanshahi M, Saleem T, Ho AK, Fuller R, Dirnberger G. A preliminary investigation of the running digit span as a test of working memory. Behav Neurol. 2008;20(1-2):17-25. doi: 10.3233/BEN-2008-0212. PMID: 19491471; PMCID: PMC5452479.
2. Blackburn HL, Benton AL. Revised administration and scoring of the Digit Span Test. J Consult Psychol. 1957;21(2):139-43. doi: 10.1037/h0047235.
3. Topp CW, Østergaard SD, Søndergaard S, Bech P. The WHO-5 Well-Being Index: a systematic review of the literature. Psychother Psychosom. 2015;84(3):167-76. doi: 10.1159/000376585. PMID: 25831962.
4. Sischka PE, Costa AP, Steffgen G, Schmidt AF. The WHO-5 well-being index: Validation based on item response theory and the analysis of measurement invariance across 35 countries. J Affect Disord Rep. 2020;1:100020. doi: 10.1016/j.jadr.2020.100020.
5. Lin M, Bieda A, Margraf J. Short form of the Sense of Coherence Scale (SOC-L9) in the US, Germany, and Russia: Psychometric properties and cross-cultural measurement invariance test. Eur J Psychol Assess. 2020;36(5):796-804. doi: 10.1027/1015-5759/a000561.
6. Hermann K, Kraus K, Herrmann K, Joos S. A brief patient-reported outcome instrument for primary care: German translation and validation of the Measure Yourself Medical Outcome Profile (MYMOP). Health Qual Life Outcomes. 2014;12:112. doi: 10.1186/s12955-014-0112-5. PMID: 25927343; PMCID: PMC5011785.
7. Baptista AF, Baltar A, Okano AH, Moreira A, Campos ACP, Fernandes AM, et al. Applications of non-invasive neuromodulation for the management of disorders related to COVID-19. Front Neurol. 2020;11:573718. doi: 10.3389/fneur.2020.573718. PMID: 33324324; PMCID: PMC7724108.
8. Alfihed S, Majrashi M, Ansary M, Alshamrani N, Albrahim SH, Alsolami A, et al. Non-invasive brain sensing technologies for modulation of neurological disorders. Biosensors (Basel). 2024;14(7):335. doi: 10.3390/bios14070335. PMID: 39056611; PMCID: PMC11274405.
9. Sturm W, Willmes K. On the functional neuroanatomy of intrinsic and phasic alertness. Neuroimage. 2001;14(1 Suppl):S76-84. doi: 10.1006/nimg.2001.0839.
10. Weibel S, Menard O, Ionita A, Boumendjel M, Cabelguen C, Kraemer C, et al. Practical considerations for the evaluation and management of attention deficit hyperactivity disorder (ADHD) in adults. Encephale. 2020;46(1):30-40. doi: 10.1016/j.encep.2019.06.005. PMID: 31610922.
11. Kolimechkov S, Seijo M, Swaine I, Thirkell J, Colado JC, Naclerio F. Physiological effects of microcurrent and its application for maximising acute responses and chronic adaptations to exercise. Eur J Appl Physiol. 2023;123(3):451-65. doi: 10.1007/s00421-022-05097-w. PMID: 36399190; PMCID: PMC9941239.
12. Pethig R. Dielectric properties of body tissues. Clin Phys Physiol Meas. 1987;8 Suppl A:5-12. doi: 10.1088/0143-0815/8/4a/002. PMID: 3568571.
13. Funk RHW, Monsees T, Özkucur N. Electromagnetic effects—from cell biology to medicine. Prog Histochem Cytochem. 2009;43(4):177-264. doi: 10.1016/j.proghi.2008.07.001.
14. National Research Council (US) Committee on Assessment of the Possible Health Effects of Ground Wave Emergency Network (GWEN). Assessment of the possible health effects of ground wave emergency network. Washington (DC): National Academies Press (US); 1993. Chapter 6, Effects of electromagnetic fields on organs and tissues. Available from: https://www.ncbi.nlm.nih.gov/books/NBK208983/
15. Bavarian R, Khawaja SN, Ajisafe AH, Sultan AS. The efficacy of microcurrent electrical nerve stimulation in treating masticatory myofascial pain: a systematic review and meta-analysis. Cranio. 2024;42(5):555-61. doi: 10.1080/08869634.2021.2020006. PMID: 34957937.
16. Marmann P, Wiatrek W. Observational study to assess the efficacy and safety of microcurrent therapy with a portable device in patients suffering from chronic back pain, skeletal system pain, fibromyalgia, migraine or depression. Med Devices (Auckl). 2023;16:261-80. doi: 10.2147/MDER.S436667. PMID: 38090106; PMCID: PMC10712256.
17. Avendaño-Coy J, López-Muñoz P, Serrano-Muñoz D, Comino-Suárez N, Avendaño-López C, Martin-Espinosa N. Electrical microcurrent stimulation therapy for wound healing: a meta-analysis of randomized clinical trials. J Tissue Viability. 2022;31(2):268-77. doi: 10.1016/j.jtv.2021.12.002. PMID: 34903470.
18. Sischka PE, Costa AP, Steffgen G, Schmidt AF. The WHO-5 well-being index: validation based on item response theory and the analysis of measurement invariance across 35 countries. J Affect Disord Rep. 2020;1:100020. doi: 10.1016/j.jadr.2020.100020.
19. World Health Organization. Mental health: a state of well-being. Geneva: World Health Organization; 2014.
20. Paterson C, Britten N. A narrative review shows the unvalidated use of self-report questionnaires for individual medication as outcome measures. J Clin Epidemiol. 2005;58(10):967-73. doi: 10.1016/j.jclinepi.2005.01.014.
21. Paterson C. Seeking the patient’s perspective: a qualitative assessment of EuroQol, COOP-WONCA charts and MYMOP2. Qual Life Res. 2004;13(5):871-81. doi: 10.1023/B:QURE.0000025586.39478.ab.
22. Paterson C, Britten N. In pursuit of patient centered outcomes: a qualitative evaluation of the 'Measure Yourself Medical Outcome Profile. J Health Serv Res Policy. 2000;5(1):27-36. doi: 10.1177/135581960000500107.
23. Paterson C. Measuring outcomes in primary care: a patient generated measure, MYMOP, compared with the SF-36 health survey. BMJ. 1996;312(7037):1016-20. doi: 10.1136/bmj.312.7037.1016.
24. Kiresuk TJ, Sherman RE. Goal attainment scaling: general method for evaluating comprehensive community mental health programs. Community Ment Health J. 1968;4(6):443-53. doi: 10.1007/BF01530764.
25. Bös C, Gaiswinkler L, Fuchshuber J, Schwerdtfeger A, Unterrainer HF. Effect of yoga involvement on mental health in times of crisis: a cross-sectional study. Front Psychol. 2023;14:1096848. doi: 10.3389/fpsyg.2023.1096848. PMID: 37034922; PMCID: PMC10074601.
26. Danigno JF, Dias MDS, Horta BL. Sense of coherence and substance use in adults: a systematic review and meta-analysis. Cad Saude Publica. 2024;40(9):e00141323. doi: 10.1590/0102-311XEN141323. PMID: 39319946; PMCID: PMC11415044.
27. Van Breukelen GJ. ANCOVA versus change from baseline: more power in randomized studies, more bias in nonrandomized studies [corrected]. J Clin Epidemiol. 2006;59(9):920-5. doi: 10.1016/j.jclinepi.2006.02.007. Erratum in: J Clin Epidemiol. 2006;59(12):1334. PMID: 16895814.
28. Ghazisaeedi M, Mahmoodi H, Arpaci I, Mehrdar S, Barzegari S. Validity, reliability, and optimal cut-off scores of the WHO-5, PHQ-9, and PHQ-2 to screen depression among university students in Iran. Int J Ment Health Addict. 2022;20(3):1824-33. doi: 10.1007/s11469-021-00483-5. PMID: 33495691; PMCID: PMC7817067.
29. Kirsch I. The emperor’s new drugs: exploding the antidepressant myth. New York: Basic Books; 2010.
30. Finniss DG, Kaptchuk TJ, Miller F, Benedetti F. Biological, clinical, and ethical advances of placebo effects. Lancet. 2010;375(9715):686-95. doi: 10.1016/S0140-6736(09)61706-2.
31. Bech P, Timmerby N. An overview of which health domains to consider and when to apply them in measurement-based care for depression and anxiety disorders. Nord J Psychiatry. 2018;72(5):367-73. doi: 10.1080/08039488.2018.1465592. PMID: 29714082.
32. Halliday JA, Hendrieckx C, Busija L, Browne JL, Nefs G, Pouwer F, et al. Validation of the WHO-5 as a first-step screening instrument for depression in adults with diabetes: results from Diabetes MILES - Australia. Diabetes Res Clin Pract. 2017;132:27-35. doi: 10.1016/j.diabres.2017.07.005. PMID: 28783530.
33. Krieger T, Zimmermann J, Huffziger S, Ubl B, Diener C, Kuehner C, et al. Measuring depression with a well-being index: further evidence for the validity of the WHO Well-Being Index (WHO-5) as a measure of the severity of depression. J Affect Disord. 2014;156:240-4. doi: 10.1016/j.jad.2013.12.015. PMID: 24412323.
Received: March 24, 2025;
Accepted: April 22, 2025;
Published:April 28, 2025 .
To cite this article : Schmieke M, Marmann P, Healy GmbH. Efficacy and Safety of a Microcurrent Device with High-Frequency Carrier Stimulation for Enhancing Active Attention: A Randomized Sham-Controlled Pilot Trial. Health Education and Public Health. 2025; 8(2): 601-609. doi: 10.31488/HEPH.195.
© The Author(s) 2025. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
